Scuba diving is an incredible way to explore the underwater world, but to do so safely, divers must understand the physics of gases under pressure. In our Part 1, we discussed Boyle’s Law, which explains the relationship between pressure and volume. Now, in Part 2, we turn our attention to Charles’ Law, another essential gas law that governs scuba diving. Understanding Charles’ Law is not just academic—it helps divers anticipate changes in gas volume and pressure when dealing with temperature variations underwater.
What is Charles’ Law?
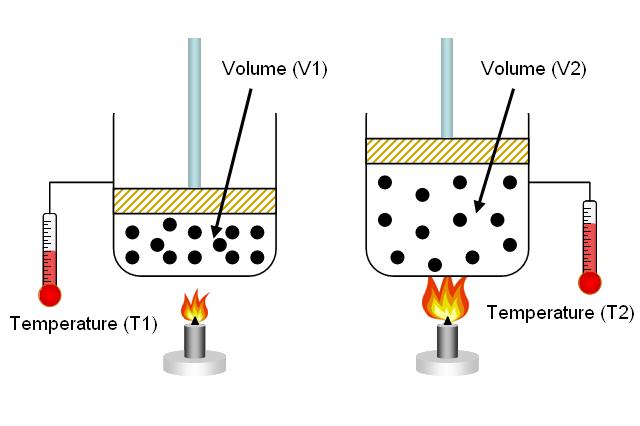
Charles’ Law, named after the French scientist Jacques Charles, describes the relationship between volume and temperature of a gas, assuming pressure remains constant.
In simple terms:
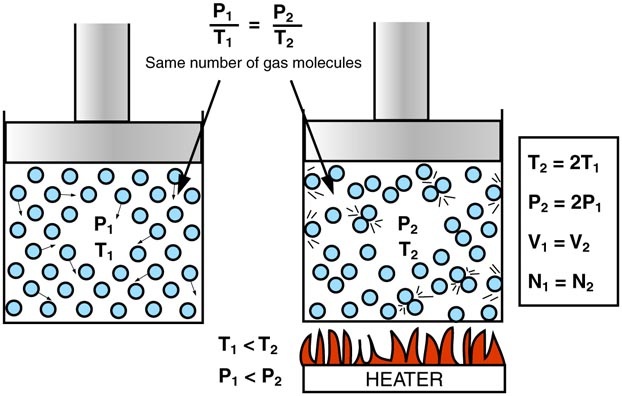
“The volume of a gas is directly proportional to its absolute temperature (in Kelvin) if the pressure is constant.”
Mathematically, Charles’ Law is expressed as:
V1T1=V2T2\frac{V_1}{T_1} = \frac{V_2}{T_2}T1V1=T2V2
Where:
- V1V_1V1 = initial volume of gas
- T1T_1T1 = initial temperature (in Kelvin)
- V2V_2V2 = final volume of gas
- T2T_2T2 = final temperature (in Kelvin)
This means that as the temperature of a gas increases, its volume increases, and as the temperature decreases, the volume decreases—provided the pressure remains constant.
Understanding Charles’ Law in Everyday Life
Before diving underwater, it helps to understand the principle above water. Some common examples include:
- Hot air balloons – When the air inside the balloon is heated, its volume increases, making the balloon rise.
- Inflating a balloon in cold vs. warm weather – A balloon expands when heated and contracts in cold weather.
- Car tires – Tire pressure and volume change with temperature, which is why checking tires on a hot or cold day can give different readings.
These examples demonstrate how gas volume responds to temperature—a principle that applies underwater too.
Charles’ Law and Scuba Diving
In scuba diving, Charles’ Law is especially relevant in gas management, buoyancy control, and equipment considerations. Let’s break it down:
1. Gas Cylinder Volume
When filling a scuba tank with compressed air, the temperature of the gas affects its volume.
- Cold tank: If a tank is filled in a cold environment, the gas volume inside is slightly lower. As the tank warms to ambient temperature, the gas expands.
- Hot tank: Conversely, a hot tank may show a higher pressure on the gauge at filling, but this decreases as the tank cools.
This is why dive shops consider temperature corrections when filling tanks, ensuring divers get the correct gas volume for their planned dive.
2. Buoyancy Considerations
Charles’ Law also explains why a BCD (Buoyancy Control Device) behaves differently with temperature changes:
- Air expands when warmed, so if you inflate your BCD in the sun, it may over-expand underwater as the water cools, affecting buoyancy.
- Conversely, if you inflate your BCD in cold water, it may compress slightly, giving less lift than expected.
Understanding this helps divers adjust buoyancy gradually and anticipate volume changes due to temperature fluctuations.
3. Dry Suit Inflation
For dry suit divers, Charles’ Law is particularly important:
- When you add air to a dry suit, warming the suit (from body heat or warmer water layers) can increase the volume of air, making the suit more buoyant.
- If you ascend to colder water layers, the air contracts, reducing buoyancy and requiring adjustment.
This temperature-volume relationship is critical for safe buoyancy management and preventing uncontrolled ascents or descents.
Temperature Effects on Dive Planning
Divers need to account for temperature when planning dives, especially in variable-depth or thermocline environments:
- Cold water dives – The gas in your tank contracts slightly, but the difference is usually minor at typical diving depths.
- Warm water dives – Gas expands more with increased temperature, and if tanks were filled in cooler air, this expansion can slightly increase pressure.
- Altitude dives – At higher altitudes, air temperature and density variations affect volume and pressure calculations, and Charles’ Law is part of the equation for accurate gas planning.
By understanding these principles, divers can make more accurate adjustments to tank fills, BCD inflation, and dry suit buoyancy.
Practical Tips for Divers Using Charles’ Law
Here are some practical applications of Charles’ Law in scuba diving:
- Check tank temperature before filling – Avoid filling tanks that are too hot or too cold without accounting for temperature correction.
- Monitor BCD inflation in varying water temperatures – Small volume changes due to temperature can affect buoyancy; adjust slowly.
- Expect dry suit air volume changes – Warm your dry suit gradually to avoid rapid expansion that could cause uncontrolled buoyancy.
- Be cautious with surface-supplied air – Gases delivered from compressors on hot days may behave differently than expected underwater.
- Plan dives with thermoclines in mind – Moving between water layers of different temperatures can slightly affect gas volume in your BCD or dry suit.
Charles’ Law vs. Boyle’s Law
It’s important to remember that Charles’ Law and Boyle’s Law complement each other, but they describe different relationships:
- Boyle’s Law – Volume vs. pressure (temperature constant)
- Charles’ Law – Volume vs. temperature (pressure constant)
Understanding both helps divers predict how gases behave under varying conditions, keeping dives safe and enjoyable.
Fun Fact: Absolute Temperature Matters
When applying Charles’ Law, always use the Kelvin scale (0 K = −273.15°C). Why? Because gas volume is proportional to absolute temperature, not the Celsius or Fahrenheit scale.
For example:
- Water temperature: 20°C → 293 K
- Water temperature: 30°C → 303 K
Even a small change of 10 K can slightly expand gas volume, which is significant for precise buoyancy adjustments in technical diving.
Safety Takeaways
Understanding Charles’ Law isn’t just theoretical—it has real-life implications:
- Always be mindful of temperature differences between tank fill and dive conditions.
- Make slow, controlled adjustments to BCD and dry suit inflation.
- Remember that volume expansion and contraction affect buoyancy, not just pressure.
- Combine knowledge of Charles’ Law with other scuba gas laws (Boyle, Dalton, Henry) for comprehensive dive planning.
By applying Charles’ Law, divers gain greater control over buoyancy, safety, and overall dive experience.
Conclusion
Charles’ Law is a foundational concept in scuba diving physics, helping explain how gas volume changes with temperature. From tank fills to BCD inflation and dry suit management, this law has practical applications every diver should understand.
While temperature changes underwater may seem minor, small differences can affect buoyancy and gas behavior, especially during longer or deeper dives. By mastering Charles’ Law alongside other gas laws, divers become more confident, precise, and safe in the underwater environment.
Next time you prepare for a dive, remember: the air you breathe isn’t just oxygen—it’s a physical system obeying the laws of the universe, and Charles’ Law is one of its guiding principles.
FAQs About Charles’s Law for Scuba Diving
Charles’ Law states that gas volume is directly proportional to temperature if pressure is constant. In diving, this helps explain how air in tanks, BCDs, and dry suits expands or contracts with temperature changes.
Gas volume in your tank can slightly expand or contract depending on the temperature at filling versus the water temperature during your dive. Dive shops often account for this to ensure accurate fills.
Yes. Warm water or body heat can expand air in your BCD, increasing buoyancy, while cold water can compress air, reducing lift. Divers should adjust slowly to maintain control.
Inflated air in a dry suit expands with warmer temperatures and contracts in colder layers. Properly managing this prevents uncontrolled ascents or descents.
Absolutely. Deep or technical dives often involve temperature changes in thermoclines, and understanding gas expansion/contraction is critical for safety.